-
PDF
- Split View
-
Views
-
Cite
Cite
Wietske Kievit, Jaap Fransen, Maarten C. de Waal Malefijt, Alfons A. den Broeder, Piet L. C. M. van Riel, Treatment changes and improved outcomes in RA: an overview of a large inception cohort from 1989 to 2009, Rheumatology, Volume 52, Issue 8, August 2013, Pages 1500–1508, https://doi.org/10.1093/rheumatology/ket166
Close - Share Icon Share
Abstract
Objective. The introduction of effective treatment strategies in the past two decades has changed the management of RA dramatically. The objective was to analyse the changes in disease activity, function, joint damage and incidence of orthopaedic surgery over a period of 20 years (1989–2009) for patients with RA.
Methods. Data acquired from 1989 to 2008 inclusive from the Nijmegen RA inception cohort were studied. By repeated measures analysis the course of the population mean disease activity score (DAS28) and the Health Assessment Questionnaire-Disability Index (HAQ-DI) corrected for age, gender, RF and disease duration was determined. Orthopaedic interventions were analysed as incidence rates with a Poisson distribution. We calculated the prevalence of the various therapies that patients were receiving.
Results. By 2009, 992 patients with RA had been included and 273 had been excluded. From 1989 onwards, the proportion of patients using MTX increased from 5% (8 of 164) to 62% (486 of 780), and biologic response modifiers from 0% to 22% (168 of 780) in 2008. The average MTX dosage increased to 16.1 ± 5.5 mg/week in 2008. The mean DAS28 (3.1) and HAQ-DI (0.47) were least (P < 0.008) in 2008 compared with previous years. There was a significant trend towards lower incidence rates of orthopaedic intervention in the period 2006–2008 than in almost all previous years.
Conclusion. Treatment strategy changed in a large inception cohort of patients with RA which coincided with decreased disease activity, increased functional ability and fewer orthopaedic interventions since the early 1990s.
Introduction
The therapeutic management of RA has changed dramatically in recent decades [1]. The effects of many of the conventional DMARDs now in regular use for the treatment of RA, such as MTX and SSZ, were discovered halfway through the 20th century. However, MTX and SSZ have only been widely used in the treatment of RA since the early 1980s [2, 3]. The introduction of combination therapies [4, 5] and tight control strategies [6–8] boosted the use of DMARDs. MTX is now regarded as the anchor drug for the treatment of RA [9]; it has been started increasingly earlier in the disease process at higher dosages [10]. Now it is common to start RA treatment with MTX as monotherapy or in combination with other DMARD [10], and the maximum dosage of MTX has increased to 30 mg/week. Since the pivotal role of TNF in the pathogenesis of RA was proved in the early 1990s, TNF inhibitors have had a central role in treating RA. TNF inhibitors can be very effective in those patients who fail on conventional DMARDs especially MTX [11–16]. After failure of the first TNF inhibitor, a second TNF inhibitor or a biologic response modifier with another mechanism of action has proved effective [17–19] and is advised [20].
These major developments of both tight control strategies and biologic agents over calendar time in the drug treatment of RA should have their impact on important outcomes such as disease activity, functional disability, joint damage, need for joint replacement and, ultimately, quality of life. A trend towards better control of RA has recently become evident in separate reports, which show decreasing disease activity, joint damage and functional disability [21–24]. Most of the previous articles report on the follow-up of patients in different cohorts defined by year or period of disease onset. However, when studying the effect of changes in treatment over calendar time on outcomes, one should also analyse those outcomes over calendar time. Furthermore, none of the previous articles reported simultaneously on several outcomes that are relevant for patients with RA covering the pre-biologic as well as the biologic era. The objective of this study was to analyse the changes in disease activity, function, joint damage and incidence of orthopaedic surgery over a period of 20 years (1989–2009) for patients with RA.
Patients and methods
Design
The Nijmegen early RA cohort started in 1985 and is an inception cohort that includes patients in two independent clinics in The Netherlands: the Department of Rheumatology of the Radboud University Nijmegen Medical Centre (RUNMC) and the Sint Maartenskliniek (SMK) in Nijmegen. The two hospitals are the main providers of RA care for the whole region, which minimizes selection bias. Inclusion in the cohort started in 1985 in the RUNMC and in 1991 in the SMK, and new patients are still being included. In The Netherlands, general practitioners do not treat RA patients themselves but refer all patients with suspected RA to rheumatologists for diagnosis and treatment, and all the rheumatologists are clinic-based. The local ethics committee (Committee on Research Involving Human Subjects Arnhem-Nijmegen) approved the Nijmegen early RA cohort protocol and patients were informed verbally and by written patient information.
Patients
Patients were consecutively included in the cohort if they fulfilled the 1987 Revised Criteria for the Classification of Rheumatoid Arthritis issued by the ACR [25], had a disease duration <1 year, had not been treated with DMARDs or steroids in the previous 4 weeks and gave their informed consent. After 2009, patients who did not fulfil ACR criteria but were considered to have RA according to the rheumatologist were included as well. All patients were treated at the discretion of their rheumatologists. Patients were followed up until death or until refusal to participate further. A more detailed description of the Nijmegen RA inception cohort is given elsewhere [26].
Assessments
The assessment protocols were the same for both clinics, and patients had assessments at 3-month intervals in the first 3 years and at least every 12 months thereafter. Trained research nurses assessed disease activity using the disease activity score (DAS28). The DAS28 includes a 28-joint count for swelling, a 28-joint count for tenderness, ESR and a patient global assessment on visual analogue scale [27]. The patients filled in a Dutch version of the Health Assessment Questionnaire-Disability Index (HAQ-DI) [28, 29] at baseline and every 6 months thereafter.
Plain radiographs of hands and feet were taken at baseline, annually in the first 3 years, and every three years thereafter in the RUNMC cohort. Radiographs of hands and feet were rated in chronological order, according to the Ratingen score using reference pictures [30]. The Ratingen score (range 0–190) is a modification of the Larsen score and evaluates joint surface destruction, graded from 0 to 5 for 38 hand and foot joints. The inter-rater reliability between the four raters was good, with an intraclass correlation coefficient of 0.85, which was tested using radiographs for 10 patients over 9 years.
All outcome measures were entered prospectively in a database that also contained information concerning age, gender, RF positivity, date of symptom onset, medical treatment and orthopaedic interventions. The orthopaedic interventions included were arthrodeses, prostheses (primary and revisions), carpal tunnel releases, orthopaedic removal procedures such as synovectomies, arthrotomies or osteotomies, nodule resection and joint or bone reconstructions, all classified as being primarily for the treatment of RA-related sequelae or complications.
Statistical analyses
The objective of this study was to analyse the changes in disease activity, function, joint damage and incidence of orthopaedic surgery over a period of 20 years (1989–2009) for patients with RA. Therefore, all outcomes, where appropriate, were analysed using repeated measures analysis, with the outcome as dependant variable and calendar year as independent variable. Because age, gender, RF and disease duration can be prognostic factors for the course of RA, we chose to include them as potential confounders in all analyses. In a further attempt to maximally control for disease duration as a confounder, data collected in the period 1985–1988 were excluded from the analyses because the cohort consisted mainly of newly diagnosed patients at that time. If those early years were to be included in the analyses, early RA patients would be heavily overrepresented in those years and outcomes are to a large extent the results of therapy initiation after diagnosis.
For the DAS28 and HAQ as outcomes, the patient-averaged DAS28 and HAQ-DI were calculated for each calendar year. The course over calendar time was analysed by repeated measures analysis including confounder correction (mixed model procedure).
For analysis of the course of the Ratingen score, we selected patients with a 3-year duration of disease from each calendar year, because generally, most joint damage progression occurs in the first 3 years of RA [31–33]. Progression in joint damage was calculated as the difference in Ratingen score after a disease duration of 3 years and the Ratingen score at diagnosis, and changes in progression rate over calendar time were analysed using repeated measures analyses (mixed models) again corrected for all above-mentioned confounders. Similarly, the proportion of patients with joint damage (Ratingen score > 0) at diagnosis and at a disease duration of 3 years was calculated. The presence of joint damage in both groups of patients separately was analysed with logistic regression analyses corrected for all above-mentioned confounders.
Generalized linear models with a binomial distribution were used to assess the occurrence of RA-related orthopaedic surgeries, as the number of occurrences in each calendar year divided by the patient time in each calendar year. Again we included confounders (age, gender, disease duration and RF) in the analyses.
In order to analyse to what degree different treatment options were used in each calendar year, the prevalences of patients being treated with MTX monotherapy, SSZ monotherapy, any combination of DMARDs and a biologic response modifier were calculated and plotted for each calendar year. Time from diagnosis to initiation of the first biologic response modifier was analysed for differences between calendar years. The mean dose of MTX per year was plotted to analyse a possible trend in the dose of MTX apart from the increased proportion of patients using MTX. Missing data were considered as missing at random and were not addressed in a specific manner. We used SPSS 16.0 for Windows for all analyses.
Results
Patients
On 1 January 1989, 136 RA patients had been included in the cohort; on 1 January 2009, 992 RA patients had been included, and the total follow-up was 8419 patient-years. By the end of 2008, 273 patients (28%) were lost to follow-up for various reasons: 97 (10%) were no longer willing to participate in the data collection, 81 (8%) had died, 60 (6%) had severe comorbidities preventing participation, 19 (2%) had moved to another area, in 4 (0.4%) patients the diagnosis of RA could not be confirmed after careful review and for 12 (1%) the reason was not known. Data of these patients were included in the analysis until the moment of lost to follow-up. Supplementary Table S1 available at Rheumatology Online gives an overview of the number of patients included every year as well as the descriptives of the population in each calendar year.
The proportions of patients with positive RF (ranging from 75% to 80%), the proportions of female patients (ranging from 62% to 67%) and the median number of months from symptom onset to diagnosis did not differ between calendar years. Average age and average disease duration (time from diagnosis) increased over calendar years from 56 ± 14.5 years in 1989 to 61 ± 13.3 years in 2008 and from 1.9 ± 1.8 years in 1989 to 7.9 ± 6.1 years in 2008, respectively.
Treatment
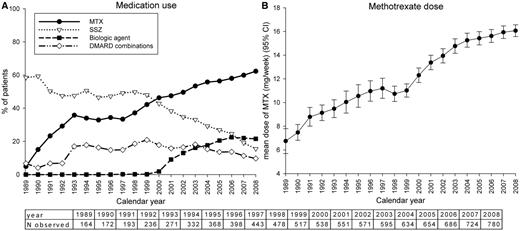
Fig. 1A shows the proportion of patients who received different treatments. The proportion of patients being treated with SSZ decreased over time, which coincided with an increase of the proportion of patients being treated with MTX up to 62% (475 of 780) in 2008. Since 1989, the proportion of patients treated with a combination of conventional DMARDs increased to 21% (108 of 517) in 1999, but decreased again to 10% (76 of 780) in 2008 (Fig. 1A). From the start of the biologic era in 1999, the proportion of patients being treated with a biologic response modifier increased to 22% (168 of 780) at the end of 2008.
Proportions of patients receiving different drug treatments (A) and the mean MTX dose (B) over time.
There was no clear trend showing that time from diagnosis to administration of the first biologic agent decreased as the years passed: median and interquartile range (P25–P75) 6.4 years (2.4–9.7) in 2001 and 5.7 years (2.6–9.7) in 2008. The MTX dose increased significantly from 6.7 ± 1.3 mg/week with a maximum of 7.5 mg/week in 1989 to 16.1 ± 5.5 mg/week with a maximum of 30 mg/week in 2008 (Fig. 1B). The mean dosage of oral prednisone was 10 mg/day in 1989; it decreased steadily and remained constant at a mean of 5 mg/day from 1999 to 2008 inclusive. The proportion of patients who used oral prednisone was fairly constant over time: it was lowest at 12% (66 of 517) in 1999 and highest at 21% (36 of 172) in 1990.
Disease outcomes
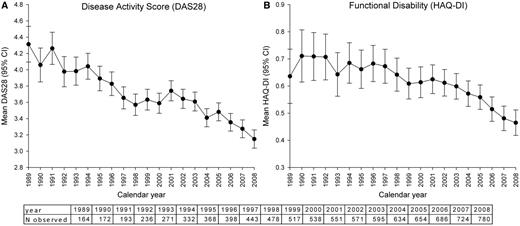
Fig. 2A shows the mean DAS28 and Fig. 2B shows the HAQ-DI by calendar year conditional on disease duration, age, gender and RF. According to the repeated measures analysis, both the mean DAS28 and the mean HAQ-DI in the RA population significantly decreased (P < 0.0001 for both).
Mean disease activity (A) and functional disability (B) over time.
The mean scores are conditional on age, gender, RF and disease duration.
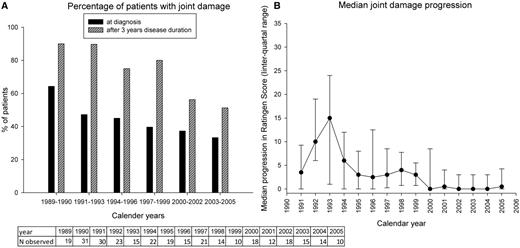
Ratingen joint damage scores were available for 306 patients until 2005. Fig. 3 shows the prevalence of joint damage and the population median progression in Ratingen score for patients with a 3-year disease duration. The graph shows that joint damage at diagnosis and after 3 years of disease is less in recent calendar years than in the early 1990s. Logistic regression analysis confirmed that the prevalence of joint damage decreased significantly (P < 0.05) over calendar years. The repeated measure analysis shows that the joint damage progression also decreased significantly (P < 0.05) over time.
Joint damage (A) and mean Ratingen score (B) for patients with a 3-year disease duration.
Orthopaedic surgery
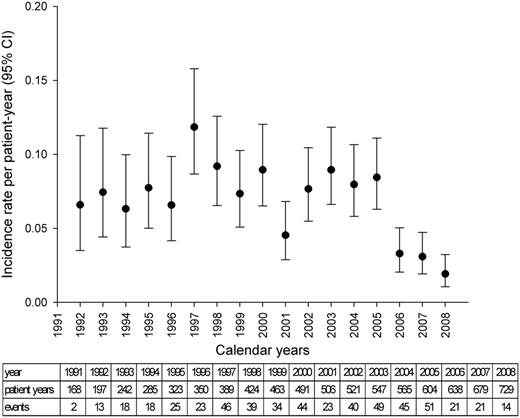
The orthopaedic surgeries were registered in the database from 1991. Fig. 4 shows the incidence rate per patient-year plotted for all subsequent years. There was a trend towards lower rates of orthopaedic surgery in the years 2006–2008 than in all previous years. Regression analyses corrected for confounders (P < 0.01) confirmed this. The incidence rate was 0.02 per patient-year in 2008, which is the result of 14 surgeries registered in 729 patient-years.
Discussion
We used data acquired from 1989 to 2009 in an RA inception cohort including 992 newly diagnosed RA patients with more than 8000 patient-years of data to analyse the course of disease activity, functional disability, joint damage and orthopaedic surgeries of patients with RA. According to the analyses, there have been major improvements in RA outcomes in the last 20 years, coinciding with changes in treatment approach.
From the beginning of the 1990s, there has been a decrease in disease activity, functional disability and joint damage antedating the introduction of biologic agents. Ultimately, the number of orthopaedic surgeries decreased. Contemporaneously, the proportion of patients receiving MTX and the dose of MTX increased, in parallel to the development of the role of MTX as the anchor drug in the management of RA. A substantial proportion of patients received a biologic response modifier (20% in 2008), but we would like to emphasize that this proportion is still a minority. Although the relationship of changed treatment approach to the trend towards better disease control cannot be interpreted as a causal relationship, the most plausible explanation lies in the use of MTX for a greater proportion of patients in higher dosages, combination of DMARDs and the use of biologic response modifiers for patients who did not respond to conventional DMARDs.
Similar studies do not show all the outcomes we analysed or used, and they restricted their observation periods to one decade. In a previous study from our own group by Welsing et al. [22], 5-year cohorts were analysed: it was shown that, for patients who were diagnosed between 1995 and 1999, the DAS28 was lower at diagnosis and the decrease after diagnosis was larger than for patients diagnosed in the periods 1985–1989 and 1990–1994. We suggested that this trend of better disease control was the result of more aggressive treatment. However, we did not analyse therapy in relation to improvements in other outcome measures. Uhlig et al. [21] present results of improved health status derived by assessing data from mailed quality of life surveys in 1994, 1996, 2001 and 2004. They suggest that this improvement is due to better access to more aggressive therapies; however, they do not show treatment data. Sokka and Pincus [10] showed that early use of MTX increased from <5% in 1980 to >90% in 2008. This coincided with substantially improved outcomes measured by HAQ in one of the included centres and by Larsen radiographic scores in the other included centre. Bergström et al. [34] showed that a 1995 RA population had lower disease activity and almost twice as many DMARDs per patient compared with a 1978 RA population; however, this study does not include the biologic and tight control era. A Norwegian research group also report a reduction in orthopaedic surgery among patients with chronic inflammatory joint disease [35]. In contrast, Sokka et al. [24] show a stable occurrence of knee and hip replacement in a Finnish cohort between 1986 and 2003. The lag time between decreasing disease activity and decreasing joint damage could explain why we observed a decrease in orthopaedic surgery only since 2006. It could also explain why Sokka et al. [24] report a stable occurrence: the period ended in 2003. Our study adds important knowledge to the existing literature because we report simultaneously on disease activity, disability, joint damage, orthopaedic surgery and treatment in one large cohort over an extended period of 20 years in which RA patients were followed up from diagnosis on a regular basis. Our study covers 10 years of the pre-biologic era and 10 years of the biologic era.
Results showing whether there is a trend of increasing quality of life or increasing work participation would have been a valuable addition to our results, but such data were not available for our cohort. However, the interpretation of trends in work participation, as well as quality of life values over extended periods of time, is particularly difficult because changes in socioeconomic circumstances also influence work participation and quality of life. Because DAS28, HAQ-DI and joint damage predict the quality of life [36] and work participation [37], and because our results are consistent for all the disease outcomes we analysed, we assume that this positive trend will ultimately result in better quality of life and more work participation for patients with RA.
The question is whether mortality and (co)morbidity also improve over calendar time. Interestingly, it is reported that RA mortality does not change, and while the mortality of the general population improves this has led to the conclusion that the mortality gap is widening [38, 39]. On the other hand, an improvement in mortality rates has also been found [40]. The main causes of death reported in these studies were cardiovascular disease (CVD), and targeting CVD may improve survival in RA [41]. Intensified treatment might reduce CVD and increase survival in RA because (i) both mortality and CVD are associated with disease severity; (ii) a good response to MTX treatment lowers the rate of CVD [42, 43] and (iii) anti-TNF treatment is associated with lower rates of CVD [44–46]. However, an intensified treatment could also have a price due to an increased rate of severe adverse events. Although MTX is considered very safe, for anti-TNF treatment small though increased infection rates and malignancies have been reported [47].
The unique characteristic of our cohort is that it started including consecutive RA patients upon diagnosis in 1985, and every member of the cohort has been followed up carefully, with attention to the main outcomes relevant to RA at regular intervals. Because the two participating centres cover the whole region concerning RA care, selection bias was minimized. However, this study has its limitations. First, the observed trend towards better disease control could be due to patient characteristics changing in time at the group level. Therefore, we corrected for the characteristics age, gender, RF positivity and disease duration. However, we assume that it is unlikely that a shift in outcomes over time has been caused by a change in unmeasured influential confounders, since these patient characteristics are central prognostic factors for RA. Anti-CCP was not available for most of the patients, but RF positivity in RA reflects anti-CCP positivity well at the group level. It is to be noticed that not all included confounding factors did show large differences between calendar years, but we chose to include them in all analyses because they can be considered as prognostic factors in general. By correcting for these factors we may have lost some statistical power, but it does not invalidate the results. Second, the trends towards better RA outcomes may be attributable to a secular trend towards milder disease [48, 49], but a recent report disputes this. The report shows a causal relationship between the trends towards better RA control and more effective anti-rheumatic treatment instead of a secular trend towards milder disease [50]. An earlier referral of patients with RA-related complaints to the rheumatologist (lead time bias) could also be an explanation for the trend towards better disease control. However, we corrected all analyses for the disease activity level at diagnosis, the time between onset of symptoms and diagnosis did not decrease over time, and the time from diagnosis to treatment was short for all calendar years by design.
Although the relationship of changed treatment approach to the trend towards better disease control cannot be interpreted as a causal relationship and a secular trend cannot be excluded, we believe that the most straightforward explanation for better RA outcomes over time is intensified treatment with DMARDs and the use of biologic response modifiers. The increased use of tight control strategies in the treatment of RA has contributed to enhanced utilization of traditional DMARDs [7, 8]. We assume that tight control of treatment for RA will become mainstream in the near future [6, 51]. On the patient and population levels, there is still room for improvement in RA outcomes.
MTX has been better utilized in the treatment of RA over the past 10 years.
RA outcomes are greatly improved with even less orthopaedic surgery compared with the 1980s and the 1990s.
At patient and population levels, there is still room for improvement in RA outcomes.
Acknowledgements
We are indebted to R. Donders for advice regarding the statistical analyses, J. Fransen, M. Creemers, E. van Ede and M. Blom for scoring the X-rays, C. Vogel, J. Alberts-Pijnenborg, J. Deenen en E. de Groot-Vos for clinical data collection and T. van Gaalen, C. Versteegden, L. Schalkwijk, M. de Lange and E. Brummelkamp for data management. Conception and design: W.K., J.F., P.v.R.; data collection: M.d.W.M., A.d.B., P.v.R.; analysis: W.K.; interpretation of data: W.K., J.F., M.d.W.M., A.d.B., P.v.R.; drafting the article: W.K., J.F., P.v.R.; revising it critically: M.d.W.M., A.d.B.; final approval: W.K., J.F., M.d.W.M., A.d.B., P.v.R. Data collection in the Nijmegen inception cohort was partially funded by the Dutch Arthritis Association. There was no specific funding for writing the manuscript.
Disclosure statement: The authors have declared no conflicts of interest.
Supplementary data
Supplementary data are available at Rheumatology Online.




Comments